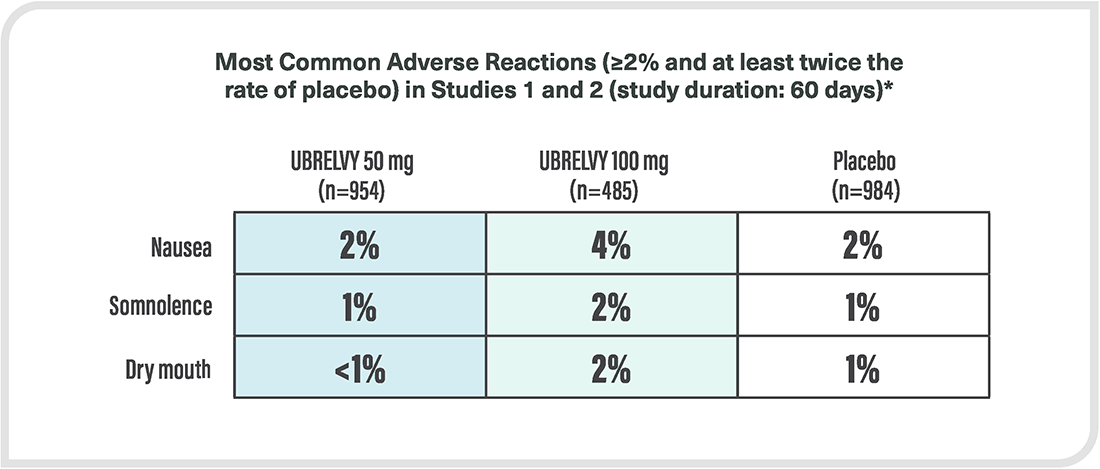
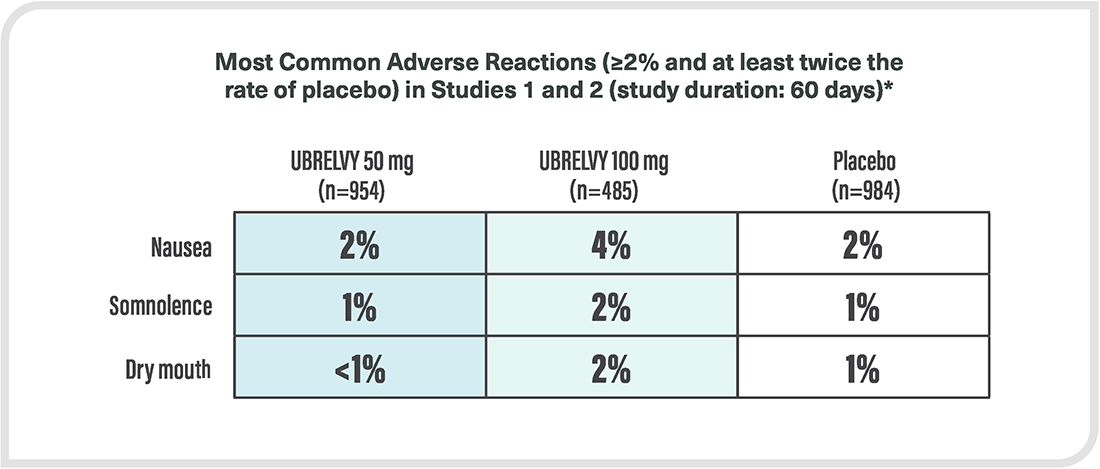
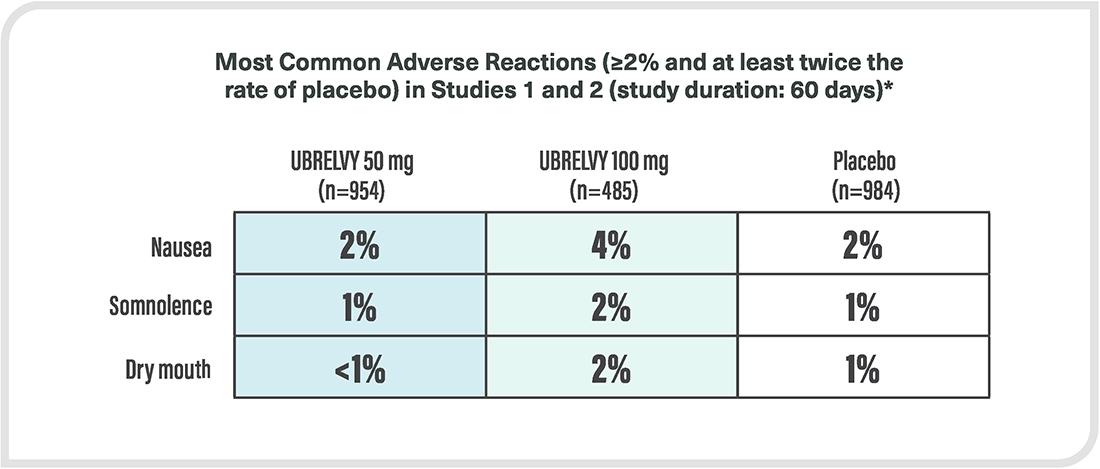
*Safety evaluated in 3664 patients who received at least one dose of UBRELVY. In the two randomized, double-blind, placebo-controlled, phase 3 trials in adult patients with migraine (ACHIEVE I and ACHIEVE II), a total of 1439 patients received UBRELVY 50 mg or 100 mg.
†Long-term safety assessed in 813 patients, dosing intermittently for up to 1 year in an open-label extension study. Patients were permitted to treat up to 8 migraines per month with UBRELVY. A total of 421 patients were exposed to 50 mg or 100 mg of UBRELVY for at least 6 months, and 364 patients were exposed to these doses for at least 1 year, all of whom treated at least 2 migraine attacks per month, on average. In this study 21,454 migraines were treated with UBRELVY.
‡Usual care patients were instructed to treat their migraine with the medication(s) that they routinely take to relieve a migraine attack.
Reference:
1. UBRELVY (ubrogepant) Product Monograph. AbbVie Corporation.